Starter Course Videos
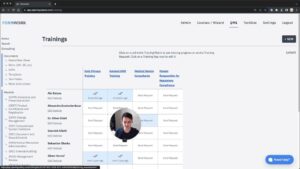
ChatGPT For Medical Devices And Automating QMS Trainings
The third (and last) way to save a lot of time is to use LLM features and to automate your…...
October 3, 2023
Auto-Generate Audit Spreadsheets For Requirements Management
When it comes to medical device compliance, where else can you save time and automate stuff? In your technical documentation…...
October 3, 2023
Save Time By Copying OpenRegulatory Templates
Here's what you can do next and save a lot of time: Use our free templates and free eQMS software,…...
October 3, 2023

How To Get Regulatory Work Done Fast: Hiring, eQMS Software, Consultants
Now you know the basics about medical device compliance. But what's next? Here's what you should do and what I…...
October 3, 2023
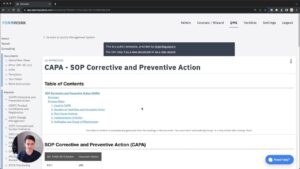
What Do You Need To Do? Looking At IEC 62304 And Other Docs
Next, let's look at what the hell "medical device documentation" actually includes. I'll walk you through our templates and give…...
October 3, 2023
Medical Device Compliance: Cost & Duration
Are you a startup founder and wondering what the hell this whole "medical device compliance" thing is about? Look no…...
October 3, 2023